Relevant Articles
Foaming power of facial cleanser = cleaning power?
29 February, 2024

"Pie Pai Gang ~ Bubble, bubble, bubble!"🫧🧙🏻♀️
Do your household facial cleansing products have foaming properties or non-foaming properties? What would you choose?
In fact, the amount of foam in a cleansing product ≠ the cleanliness. The two are not necessarily directly proportional, and strong cleaning ability may not necessarily be beneficial to the skin.
Choose a cleansing product that suits your skin, and pay close attention to the surfactant ingredients and overall formula used.
The key to a cleansing formula that is both gentle and has sufficient cleansing power is to use low-irritation surfactants, and then accurately adjust the proportions of various surfactant ingredients in the formula to improve the gentleness of the entire cleansing formula. No loss of cleanliness.
Do you think the edge is washed clean?
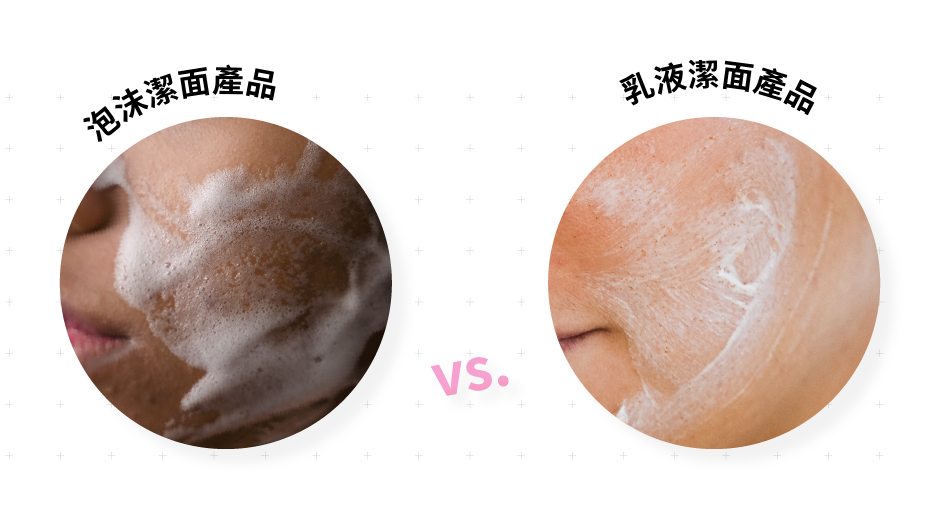
To judge the cleansing ability of a cleansing product, you don’t just rely on the amount of foam, you also need to consider the proportion of surfactants in the product.
surfactant
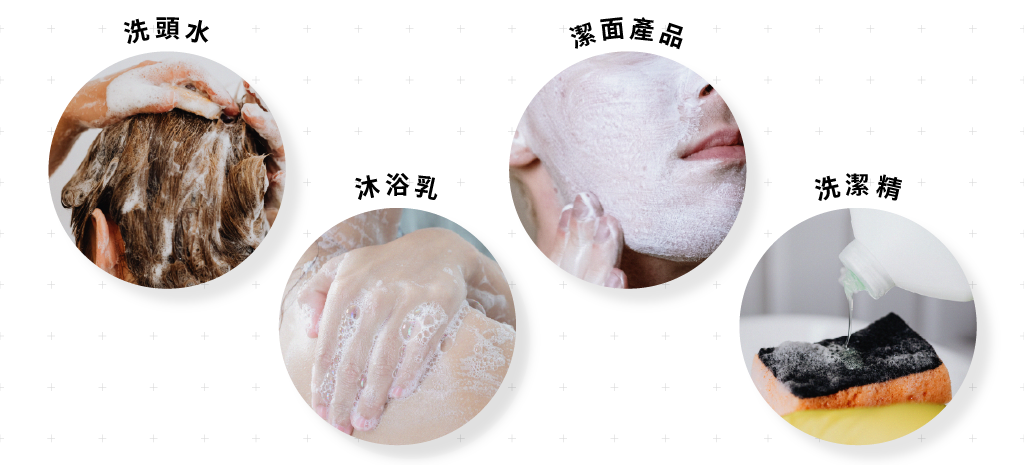
Most of the cleaning products you come into contact with daily use surfactants as the main cleaning ingredients. The type and usage ratio of different surfactants will affect the cleaning power and foaming power of the product.
Properties of surfactants
1. Reduce surface tension
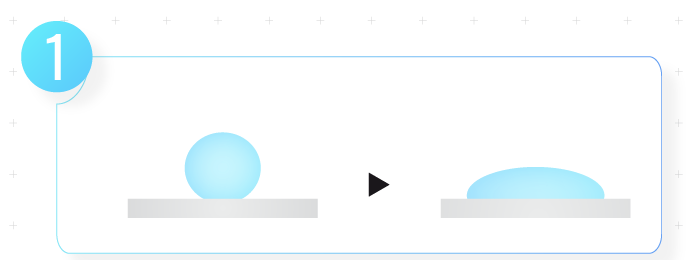
2. Hydrophilic head and lipophilic tail

3. Integrate two different interfaces

The cleaning principle of surfactant
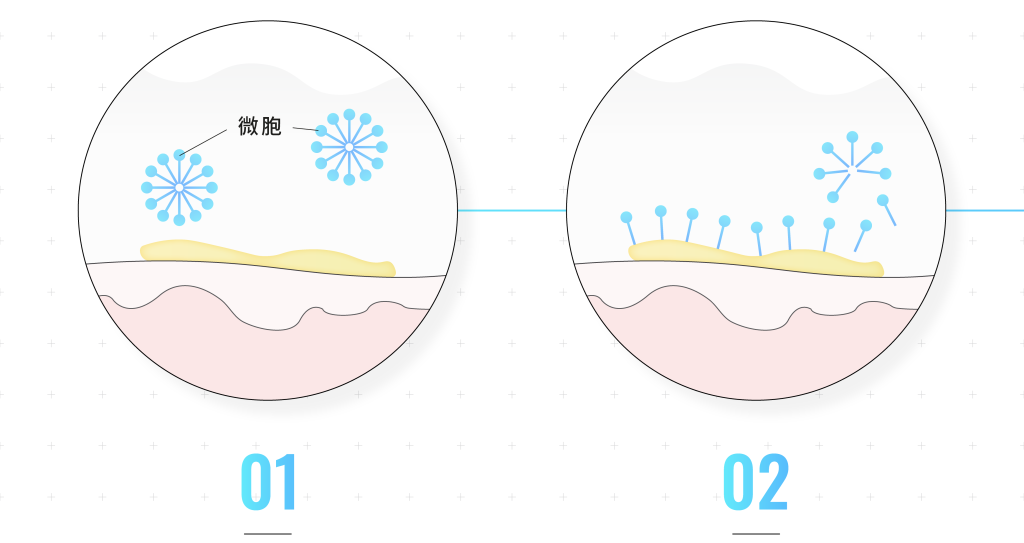
When surfactants are dissolved in water, the lipophilic ends attract each other, causing the molecules to assemble together to form micelle.
When the microcells come into contact with dirt on the face, the lipophilic end will stick to the dirt.
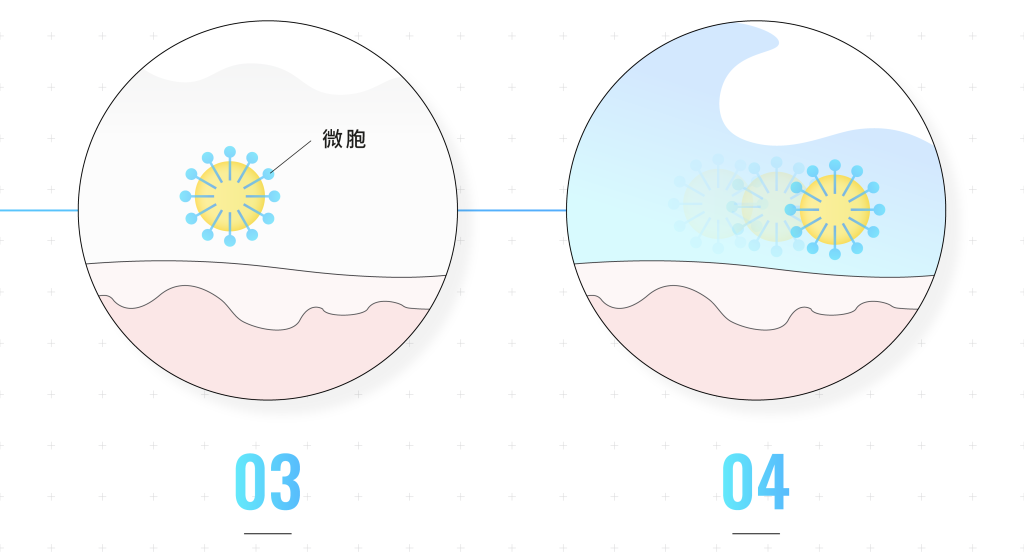
After dirt leaves the skin, it is surrounded by surfactants and reorganizes into microcells.
The microcells with the hydrophilic end facing outward will be washed away with water to achieve a cleaning effect.
How do bubbles form?
Step 1.
When a surfactant is dissolved in water, the lipophilic end will leave the water and enter the air.
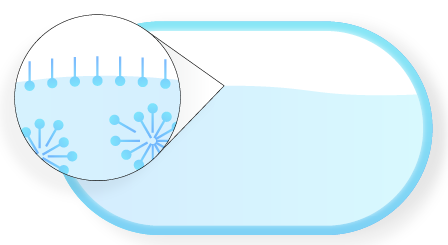
Step 2.
When external force is applied (stirring, rubbing, or using a foaming sponge), air is introduced into the solution.

Step 3
Surfactants can fuse the air and water interfaces, and the lipophilic end will enclose the air and form foam.
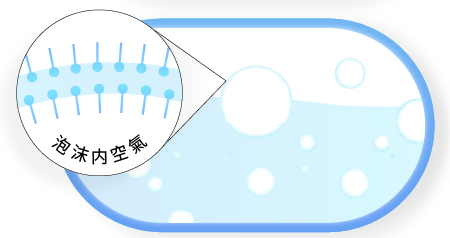
How to increase bubbles?
To know how to increase foam, you first need to understand the two main factors that affect foam quantity: foamability and stability.
Foamability
Refers to the quantity and speed of foam generation. Adding a foaming agent to the solution or using a foaming tool can effectively bring air into the solution and produce more foam.
StabilityStability
Refers to the maintenance time of the bubble. Adding foam stabilizer to the solution will make the foam structure stronger and increase the foam maintenance time.
Therefore, improving the foaming and stability of the solution can increase the number of foams, but it does not mean that the cleaning ability is also improved at the same time.
Cleaning is not about foam!

Foam is just for visual and tactile enjoyment
The cleaning ability mainly relies on surfactants, and foam is caused by the fusion of too much surfactants with air. The more foam there is, it may just reflect the high concentration of surfactants added to the product, but it does not fully reflect the effectiveness of the cleaning function.
